Project description SaAr in vitro (Schnelle anwendungsorientierte Analyse robuster in vitro Systeme)
Project objective and summary:
SaAr in vitro has the ambitious goal of displaying human inflammatory diseases using cellular in vitro models in order to test new drugs and chemicals on a scale accessible to industry. The project is based on two key components: the application-oriented mass spectrometric analytical method and human cellular in vitro models. The combination of both techniques enables the targeted development of new drug formulations for humans and the early identification of risks without the need for animal testing. The project focuses on inflammatory diseases in humans. Key targets include bronchial asthma, COPD, Crohn’s disease, and the inflammatory skin condition psoriasis. For this project, PharmBioTec draws on its own extensive expertise in the field of the 3Rs (Replacement, Reduction, and Refinement of animal testing).
The so-called omics technologies—which include the proteomics technology based on mass spectrometry used in this project—enable PharmBioTec to simultaneously and cost-effectively measure various inflammatory signals from skin, intestinal, and lung models. This application enables in-depth, high-throughput analyses at cost-effective conditions. This combination of human cell models and modern measurement technology is more cost-effective and efficient for the chemical and pharmaceutical industries than the use of animal models and offers the Saarland a new opportunity in the field of contract research.
Funding of the research project:
The project SaAr in vitro is funded by the program „EFRE Saarland im Ziel Investitionen in Beschäftigung und Wachstum 2021-2027“ with the grant reference number EFRE-AuF-0000889. The funding period runs from May 1, 2024, until July 31, 2027.



First results:
- Proteomic profiling of macrophages: effects of inflammatory activation and anti-inflammatory treatment with IBD therapeutics (Lichtner et al., 2025)
In an initial study, we investigated changes in the proteome of MDMs (macrophages derived from human monocytes) that were stimulated with lipopolysaccharide (LPS) to model an inflammatory response. Both intracellular and extracellular protein expression patterns were analyzed using HPLC-MS/MS. LPS stimulation activated key pro-inflammatory signaling pathways, including NF-κB signaling, which was reflected in increased expression of cytokine- and adhesion-related proteins such as IL-1β, IL-8, and ICAM-1.
Within this model, the mechanisms of action of three established IBD therapies (mesalazine, prednisolone, and 6-mercaptopurine) were systematically investigated. Mesalazine caused a moderate modulation of inflammatory regulators, while prednisolone triggered a significant suppression of proinflammatory and complement proteins. 6-Mercaptopurine led to extensive changes in ribosomal, metabolic, and apoptosis-associated proteins.
The results reveal clear differences between intracellular and extracellular protein signatures and confirm the role of activated macrophages in immune modulation. Furthermore, the observed proteomic changes reflect physiological processes in the human body. Overall, the study demonstrates that the LPS-stimulated MDM model replicates key aspects of macrophage activation in IBD and provides drug-specific proteomic signatures. This underscores the assay’s potential as a predictive model for human data and as a basis for personalized therapeutic strategies.
Read our study here: Proteomic profiling of macrophages: effects of inflammatory activation and anti-inflammatory treatment with IBD therapeutics – ScienceDirect
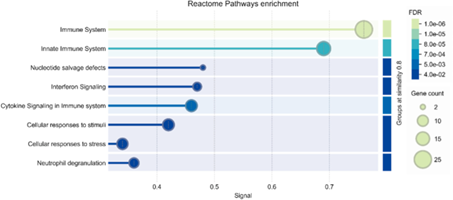
Figure 1: Reactome pathway enrichment analysis of proteins from supernatant and lysate of monocyte-derived macrophages stimulated for 24 h with LPS (positive controls) which are either significantly(negative log10 p-value of > 1.3 and a log2 ratio ≤ -1 or ≥ 1) upregulated compared to the unstimulated negative controls or were found exclusively in the LPS-stimulated samples.
Selection of varoius presentationsof the project SaAr in vitro:
- MaxQuant summer school 2024: Poster: „SaAr in vitro: Innovating Drug Testing with Human Co-Culture Cell Models and Mass Spectrometry“
- EUSAAT congress, 2024: Poster: “Investigation of complex in vitro models using shotgun proteomics for the prediction of human-relevant data for
- anti-inflammatory drugs”
- 3R Conference Saarbrücken, 2024: Talk: “SaAr in vitro – Advancing Drug Safety and Efficacy with in vitro Models and High-Throughput Proteomics in inflammatory diseases”
- HTCR Symposium, 2025: Poster: “Proteomic analyses of human-derived macrophages predict human data for anti-inflammatory drugs”
- MaxQuant summer school, 2025: Poster: “Proteomic analysis of human-derived macrophages and human intestinal cells to predict human responses”
- CRS meets BioBarriers conference 2026: Poster: “SaAr in vitro: Proteomics-Based Cell Culture Models for Predictive Drug Testing in Chronic Inflammation”
- Presentation of the project as part of the Innovation Tour led by Saarland’s Minister of Economic Affairs, Jürgen Barke (Forschung und Innovation – Innovationstour von Wirtschaftsminister Jürgen Barke – saarland.de)
- Publication of the first study: Simone Lichtner, Kathrin Schunck, Johanna Frey, Janina Osti, Selina Dannheimer, Sabrina Schnur, Claus-Michael Lehr, Marc Schneider, Marius Hittinger: „Proteomic profiling of macrophages: effects of inflammatory activation and anti-inflammatory treatment with IBD therapeutics“, European Journal of Pharmaceutics and Biopharmaceutics, Volume 216, 2025, https://doi.org/10.1016/j.ejpb.2025.114869.



